 In addition, the experimentally observed protein pI values were summarized in the experimental databases ( Hoogland et al., 2004 Bunkute et al., 2015). Some methods take into account the effect of the amino acids residues adjacent to the charged residues, such as aspartate and glutamate ( Cargile et al., 2008), effects of posttranslational modifications, such as phosphorylation and N-terminal acetylation ( Gauci et al., 2008), or effects of the presence of polyelectrolyte chains around proteins ( Srivastava et al., 2017). Various calculative algorithms have been developed for estimating protein pIs in agreement with experiments regardless of structural aspect ( Gasteiger et al., 2003 Cargile et al., 2004 Gauci et al., 2008 Maldonado et al., 2010 Audain et al., 2016). Notably, three-dimensional structure and pH of surrounding environment can influence ionizable groups and affect the net charge on the molecule significantly ( Russell and Warshel, 1985). It was demonstrated that the protein pI can be estimated based on a polypeptide sequence in close agreement with experimentally determined pI values ( Sillero and Ribeiro, 1989), and the focusing positions of proteins in immobilized pH gradients and two-dimentional gels can be reliably predicted from their amino acid composition ( Bjellqvist et al., 1993 Bjellqvist et al., 1994 Link et al., 1997). Thus, the integral property of a protein, such as protein pI, was supposed to result from discrete local acidic and basic pKas of amino acid side chains. Out of twenty common amino acids, two amino acids, aspartic acid, and glutamic acid, are negatively charged and three amino acids, lysine, arginine, and histidine, are positively charged at the neutral pH, as defined by their pKa values. Amino acid composition of a protein sequence primarily defines its pI, based on the combination of dissociation constant (pKa) values of the constituent amino acids.  
Hence, pI values have long been used to distinguish between proteins in methods for protein isolation, separation, purification, crystallization, etc. The protein pI varies greatly from extremely acidic to highly alkaline values ranging from about 4.0 to 12.0. 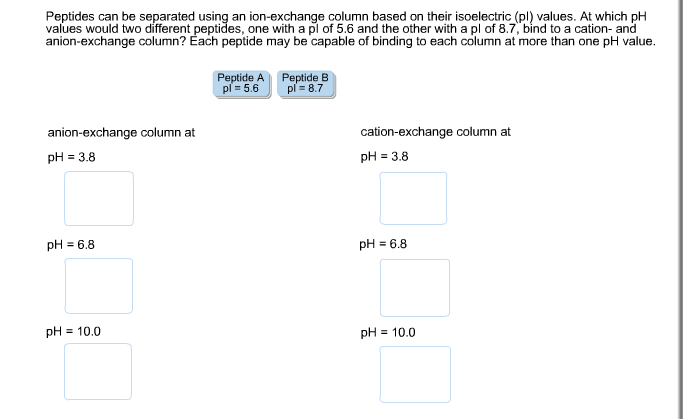
Accordingly, proteins are positively charged at a pH below their pI and negatively charged at a pH above their pI. The isoelectric point (pI) of a protein is defined as the pH at which the net charge of a protein molecule is zero. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
